 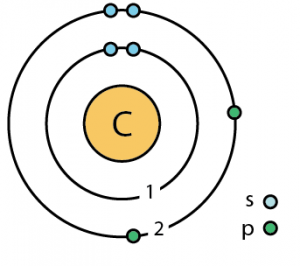 
VSEC stands for the Valence Shell Electronic Configuration This forms the first bond, the strongest of all called sigma (σ) bond. The half-filled sp(z) hybrid orbital of the carbon atom head-on overlaps with the half-filled sp(z) hybrid orbital of the oxygen atom. The below mention diagram is the valence shell electronic configuration of both the carbon and oxygen atom. The hybridization of carbon monoxide is sp as its geometrical structure is linear. Whereas, the lone pairs are drawn in the exact opposite sides where bond formation has taken place.Īs per the VSEPR theory, the electron cloud, as well as the lone pair of the electrons on both the atoms, will repel each other.Īs a result, they will be pushed apart from one another, placing both the carbon and oxygen atom 180 degrees to one another, in a linear geometry. It can further be confirmed with the help of the valence shell electron pair repulsion (VSEPR) theory, which shows the formation of a shared covalent triple bond between the carbon and oxygen atoms. It makes the geometrical structure of the carbon monoxide linear. The bond angle between the carbon and the oxygen atom is 180 degrees. Geometrical Shape of the Carbon Monoxide (CO) Choose a central atom: Both the atoms will be central.Look for the total number of bonds forming: Triple covalent bonds are forming in one carbon monoxide (CO) molecule.Find how many electrons are needed: It is 6 for one carbon monoxide (CO) molecule as per the octet rule.Find total valence electrons: It is 10 to form the carbon monoxide.The lewis structure is written in such a manner to fill the deficiency of both the atom in the case of carbon monoxide (CO).įrom the aforementioned Lewis structures of single oxygen and carbon atom, it can be analyzed that carbon and oxygen atoms have ten valence electrons which will participate in the bond formation.īelow mentioned are the steps to draw Lewis structure of Carbon Monoxide: Whereas, an oxygen atom has six valence electrons and a deficiency of two. The carbon atom has four valence electrons and a deficiency of another four. The octet rule says, the maximum number of valence electrons an atom can have in its outermost shell is eight. The electrons present in the outermost shell which is also said to be the valence electrons, of an atom participate in the bond formation with other atoms to gain stability.īecause the outermost orbit of non-noble elements is not completely filled and is unstable, the valence electrons readily react with a nearby atom to stabilize its orbit. As it needs high energy for a strong bond formation to take place.Ĭonclusion What are the Valence electrons? It is essential to realize that the larger the number of valence electrons, the more it is difficult for an atom to donate it. So, oxygen has six valence electrons in total, which are ready to act in a bond formation to stabilize its electronic configuration. On the other hand, the atomic number of an oxygen atom is eight which makes its electronic configuration 1s2 2s2 2p4.Īs the 2p shell has a capacity of holding up to six electrons in total, there is a deficiency of two electrons. So, carbon has four valence electrons which are ready to act in a bond formation to stable its atomic structure. The atomic number of the carbon is six which makes its electronic configuration 1s2 2s2 2p2.Īs the 2p shell has a capacity of holding up to six electrons, there comes a deficiency of four electrons. Lewis structure of single carbon and oxygen atom separately is as shown below. The maximum number of valence electrons that can be written around an atom is eight. The structure is drawn using dots which determine the number of valence electrons in an atom and lines between the atoms, determining the number of bonds forming. The Lewis structure, also called as electron dot structure, is a simplified method of representing the number of valence electrons present within an atom or a molecule.įurthermore, the structure helps with determining the number of lone pairs of electrons present in an atom and how they act in a bond formation. The carbon monoxide is produced from the partial oxidation of carbon dioxide (CO2) or any other carbon-containing element. 
It is so because, carbon monoxide uses hemoglobin, an oxygen carrier, to reach throughout the body when in a concentration of more than 35ppm. Carbon monoxide (CO) is a tasteless and odorless flammable gas that is quite toxic in nature to the fauna. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
